Teratogens in Pregnancy Can Cause Birth Defects
Although the embryo is well protected in the uterus, some environmental substances – teratogens – can cause developmental disorders after maternal exposure to them. Teratogen is any agent, which can cause congenital defect, or increase the frequency of defect occurrence in a population. Environmental factors, such as infections and drugs, can simulate genetic conditions, for example, when two or more children of normal parents are affected. It is important to remember that not everything that is familial is genetic.
Parts of embryos and organs most sensitive to teratogenic agents during periods of rapid differentiation (see Fig. 1). Because molecular signaling and embryonic induction precede morphologic differentiation, the period during which structures are sensitive to interference by teratogens often precedes the stage of their visible development.
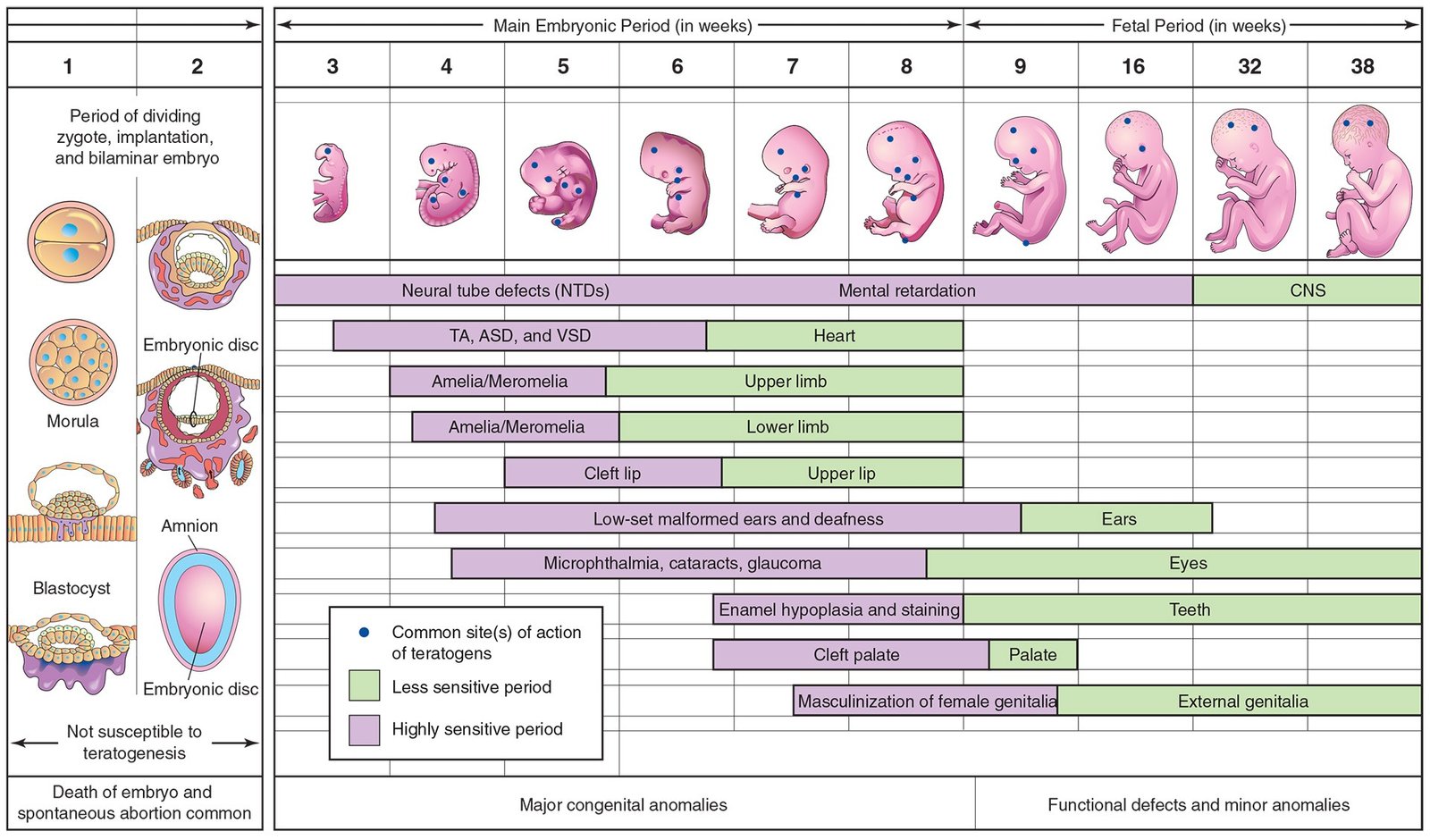
Fig. 1 Critical periods in prenatal human development. During the first 2 weeks, the embryo is usually not susceptible to teratogens. At this point, the teratogen damages all or most of the cells, which leads to the death of the embryo, or damages only a few cells, allowing the conceptus to recover, and the embryo to develop without birth defects. Purple areas indicate periods of hypersensitivity when serious defects (e.g., amelia, lack of limbs) can occur. The green sections indicate stages that are less sensitive to teratogens when minor birth defects can be caused.
Teratogens in pregnancy do not appear to be effective in causing birth defects until cellular differentiation has begun; however, their earlier actions may cause the death of an embryo. The exact mechanisms by which many drugs, chemicals and other environmental factors disrupt fetal development and causes abnormalities are unclear.
Rapid progress in molecular biology is providing additional information on the genetic control of differentiation and the cascade of molecular signals and factors controlling gene expression and pattern formation. Currently, researchers are increasingly focusing on the molecular mechanisms of abnormal development, trying to better understand the pathogenesis of congenital defects.
Principles of Teratogenesis
When considering the possible teratogenicity of an agent, such as a drug or a chemical, three factors are important to consider:
- Critical periods in prenatal human development (see Fig. 1),
- Dose of a drug or chemical,
- Genotype (genetic constitution) of the embryo.
Critical Periods of Human Development
An embryo’s susceptibility to a teratogen depends on its stage of development when an agent, such as a drug, is present. The most critical period in development is when cell differentiation and morphogenesis are at their peak. The most critical period for brain development is from 3 to 16 weeks (see Fig. 1), but its development may be disrupted by teratogens after this time, because the brain differentiates and grows rapidly at birth.
Tooth development continues long after birth; therefore, the development of permanent teeth can be impaired by tetracyclines from 18 weeks prenatal to 16 years. The skeletal system has a long critical period of development, covering childhood; therefore, skeletal tissue growth provides a good indicator of overall growth.
Environmental disturbances during the first 2 weeks after fertilization may interfere with cleavage of the zygote and implantation of the blastocyst, which may cause early death and spontaneous abortion of the embryo (see Fig. 1).
Development of the embryo is most easily disrupted when the tissues and organs are forming (see Fig. 1). During this organogenetic period (fourth to eighth weeks), teratogenic agents may induce major birth defects. Physiological defects – for example, small morphological defects of the outer ear – and functional disorders, such as a restriction of mental development, may be the result of developmental disorders during the fetal period. Each part, tissue and organ of the embryo has a critical period during which its development can be impaired (see Fig. 1). The type of birth defect depends on which parts, tissues and organs are most susceptible when the teratogen is active.
Embryologic timetables, such as the one in Fig. 1, are helpful when considering the cause of birth defects. However, it is incorrect to assume that defects always result from a single event occurring during the critical period of development, or that it is possible to determine from these timetables the day on which a defect was produced. It is known that the teratogen was supposed to disrupt the development of tissue, part or organ until the end of the critical period.
Human Teratogens in Pregnancy
Awareness that some agents may interfere with prenatal development makes it possible to prevent some birth defects. For example, if women learn about the harmful effects of drugs, alcohol, chemicals and viruses, most pregnant women avoid exposure to these teratogenic agents.
Drugs vary significantly in their teratogenicity. Some teratogens in pregnancy, such as thalidomide, cause serious developmental disorders if administered during the organogenetic period of certain parts (for example, limbs) of the embryo. Other teratogens in pregnancy cause mental and growth restrictions on embryos. Despite this, less than 2% of birth defects are caused by drugs and chemicals. Only a few drugs were positively involved as human teratogenic agents, but new agents continue to be identified. For women, it is better to avoid using all medicines during the first trimester unless there are serious medical reasons for using them.
Environmental Chemicals as Teratogens in Pregnancy
There has been increasing concern about the possible teratogenicity of environmental, industrial, and agricultural chemicals, pollutants, and food additives. Most of these chemicals were not positively associated with teratogens in humans.
Infectious Agents as Teratogens in Pregnancy
Cytomegalovirus, hepatitis B, herpes simplex, human parvovirus B19, rubella, coxsackieviruses, and viruses associated with variola, varicella, measles, and poliomyelitis may pass through the placental membrane and cause fetal infection. In some cases, hepatitis B virus can cause preterm birth or fetal macrosomia. Rubella virus can cause severe birth defects, such as IUGR, postpartum growth retardation, cardiac and major vessel abnormalities, microcephaly, sensorineural deafness, cataract, microphthalmos, glaucoma, retinopathy pigmentosa, mental deficiency, neonatal bleeding, hepatosplenomegaly, and osteoma. Treponema pallidum can cause fetal syphilis, and Toxoplasma gondii can produce destructive changes in the brain and eyes of the fetus.
Radiation as A Teratogen
Exposure to high levels of ionizing radiation can damage embryonic cells, leading to cell death, chromosomal damage, mental deficiency and inadequate physical growth. The severity of the embryonic damage is related to the absorbed dose, the dose rate, and the stage of embryonic or fetal development when the exposure occurs. Accidental exposure of pregnant women to radiation is a common cause for anxiety.
No conclusive proof exists that human congenital defects have been caused by diagnostic levels of radiation (<10,000 mrad). Scattered radiation from a radiographic examination of a part of the body that is not near the uterus (e.g., thorax, sinuses, teeth) produces a dose of only a few millirads, which is not teratogenic to the embryo. The recommended limit of maternal exposure of the whole body to radiation from all sources is 500 mrad (0.005 Gy) for the entire gestational period.
Maternal Factors as Teratogens in Pregnancy
Poorly controlled diabetes in a mother with persistent hyperglycemia and ketosis, especially during embryogenesis, is associated with a two- to three- times higher frequency of birth defects. Newborns from a diabetic mother are usually large (macrosomia). The common defects include holoprosencephaly (failure of the forebrain to divide into hemispheres), meroencephaly (partial absence of the brain), sacral agenesis, vertebral defects, congenital heart defects, and limb defects or vascular disruption.
If left untreated, women who are homozygous for phenylalanine hydroxylase deficiency – phenylketonuria – and those with hyperphenylalaninemia are at increased risk for having offspring with microcephaly (abnormal smallness of the head), cardiac defects, mental deficiency, and IUGR. The congenital defects can be prevented if the mother with phenylketonuria follows a phenylalanine-restricted diet before and during pregnancy.
Mechanical Factors as Teratogens in Pregnancy
Clubfoot and congenital dislocation of the hip can be caused by mechanical forces, especially in the deformed uterus. Such birth defects can be caused by any factor that limits the mobility of the fetus, thereby causing prolonged compression in an abnormal posture. A significantly reduced quantity of amniotic fluid (oligohydramnios) may result in mechanically induced deformation of the limbs, such as hyperextension of the knee. Intrauterine amputations or other defects caused by local narrowing during fetal growth can be the result of amniotic bands, rings formed as a result of amnion rupture in early pregnancy.












Leave a Reply
Want to join the discussion?Feel free to contribute!