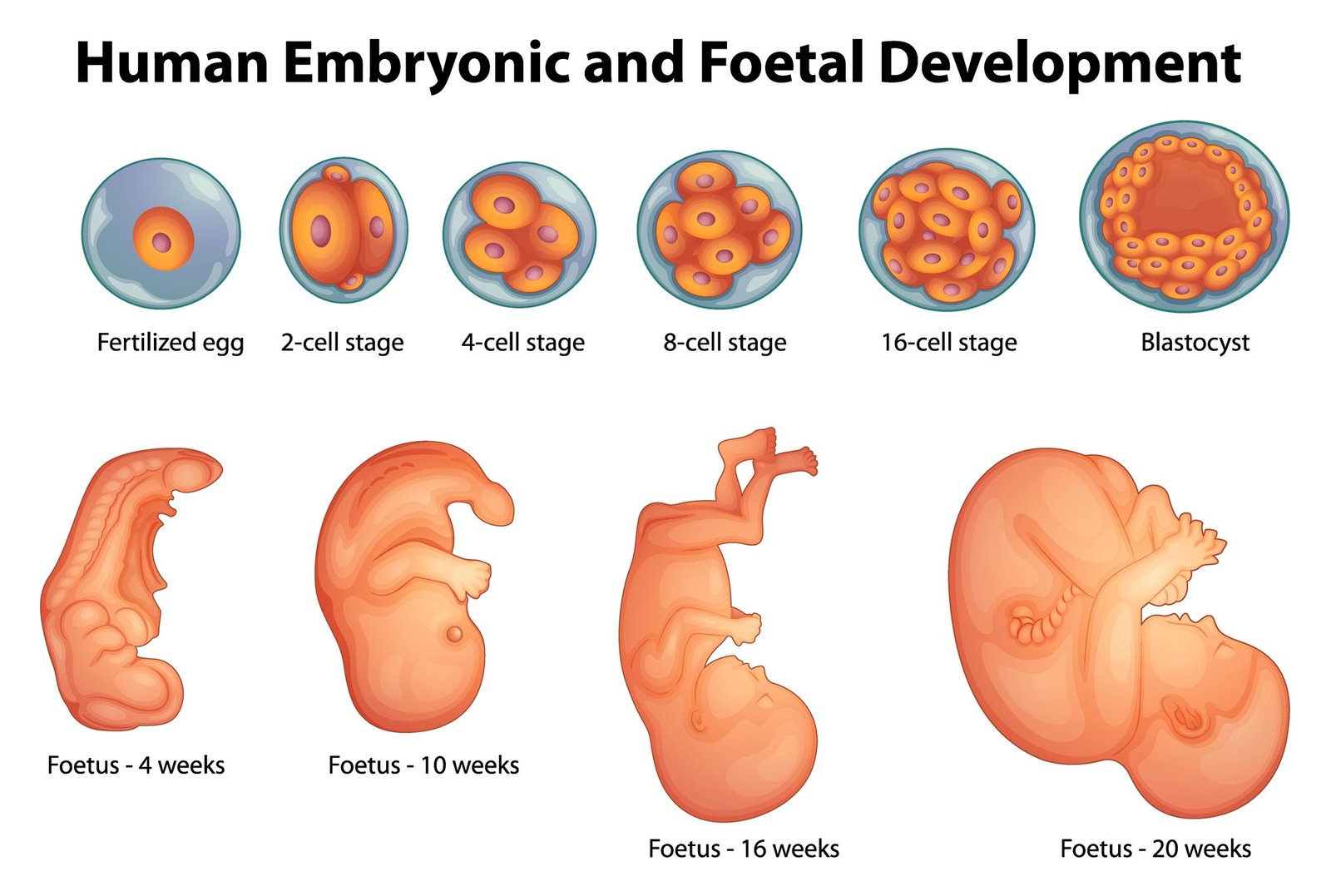
Selection of Embryos for Embryo Transfer
Historically, embryo transfer was carried out 2 days (approximately 48–54 hours) after oocyte retrieval, but transfer has been carried out from as early as 1-hour post-ICSI to 5 days later, at the blastocyst stage. Trials of zygote transfer on Day 1 also achieved acceptable pregnancy rates. It seems that the specific timing of transfer may not be crucial for the human implantation process. On Day 2, cleaved embryos may contain from two to six blastomeres. Embryo transfer 1 day later, on Day 3, or on Day 5 at the blastocyst stage is advocated as a means of selecting embryos with higher implantation potential, by the elimination of those that arrest at earlier cleavage stages in vitro.
Two major issues continue to hinder the effectiveness of ART treatment: low implantation rates and a high incidence of multiple pregnancies. Poor endometrial receptivity and adverse uterine contractions can both contribute to early embryo loss, but the low efficiency of assisted conception is widely attributed to genetic defects in the embryo.
More than 40% of ART-derived embryos are known to harbor chromosomal abnormalities. Errors in meiotic and mitotic segregation of chromosomes in the oocyte and during cleavage of early embryos can lead to various patterns of aneuploidy, including polyploidy and chaotic mosaic, which account for about one third of aneuploidy, including more than one chromosome per cell. However, despite the fact that grossly abnormal chromosome complements are lethal, in most cases the morphology of embryos that are genetically normal does not differ markedly from those with aneuploid, polyploid or mosaic chromosomal complements.
Consequently, genetically abnormal embryos after IVF or ICSI may be graded as suitable for transfer using subjective selection criteria. Developing a reliable diagnostic test that can be used to identify embryos with the greatest developmental competence continues to be a major priority in human ART, in the hope of eventually selecting a single embryo that is likely to result in a healthy live birth following transfer.
In selecting embryos for transfer, the limitations of evaluating embryos based on morphological criteria alone are well recognized: correlations between gross morphology and implantation are weak and inaccurate, unless the embryos are clearly degenerating/fragmented. Objective criteria for evaluating embryos are available in laboratories with research facilities, but may be out of reach for a routine clinical IVF laboratory without access to specialized equipment and facilities. Objective measurements of human embryo viability that have been applied historically include:
- High-resolution video-cinematography,
- Computer-assisted morphometric analysis,
- Blastomere or polar body biopsy for cytogenetic analysis,
- Culture of cumulus cells,
- Oxygen levels in follicular fluid/peri-follicular vascularization,
- Distribution of mitochondria and ATP levels in blastomeres,
- Molecular approaches:
- Metabolic assessment of culture media (amino acid profiling, metabolomics),
- Gene expression/expression of messenger (mRNA) in cumulus cells and/or embryos.
High-Resolution Video-cinematography
As early as 1989, researchers have carried out a classic experiment that aimed to clearly define morphological criteria that might be used for embryo assessment, using a detailed analysis of videotaped images. Immediately before embryo transfer, embryos were recorded on VHS for 30–90 seconds, at several focal points, using Nomarski optics and an overall magnification of x1400. The recordings were subsequently analyzed by observers who were unaware of the outcome of the IVF procedure, and they objectively assessed a total of 11 different parameters.
Nine parameters were evaluated (+) or (-), and the change in Zona thickness and the percentage of extracellular fragments received a numerical value. Analysis of these criteria showed no clear correlation with any intracellular features of morphology, but that the most important predictor of fresh embryo implantation was the percentage of variation in thickness of the zona pellucida. Embryos with a thick, even Zona had a poor prognosis for implantation.
Those whose Zona had thin patches also had “swollen,” more refractile blastomeres, and had few or no fragments. This observation was one of the parameters that led the group to introduce the use of assisted hatching. In analyzing frozen-thawed embryos, the best predictor of implantation was cell—cell adherence. The proportion of thawed embryos with more than one abnormality (77%) was higher than that of fresh embryos (38%) despite similar implantation rates (18% versus 15%).
Nearly 30 years have elapsed since these observations were published, and the quest to identify specific morphological markers of embryo implantation potential still continues—now with the help of more sophisticated technology to measure both properties of the Zona and detailed embryo morphology.
Zona Pellucida Birefringence
Polarized microscopy allows three layers to be distinguished in the zona, with the innermost layer showing the greatest birefringence (i.e., a higher level of light retardant). Several studies have investigated a possible correlation between this Zona property and implantation potential. There is no doubt that properties of the Zona may be important in assessing oocyte/embryo potential, particularly in response to exogenous FSH stimulation, but further studies are required in order to establish a clear correlation.
Computer-Assisted Morphometric Analysis
High-resolution digital images of embryos can be assessed in detail with the help of computer-assisted multilevel analysis, which provides a three-dimensional picture of embryo morphology. A system with a computer-controlled motorized stepper mounted on the microscope will automatically focus through different focal planes in the embryo to produce a sequence of digital images. Automatic calculations of morphometric information from the image sequences describe features and measurements of each embryo, including size of nucleus and blastomeres and their spatial positions within the embryo, as well as features of the zona pellucida; all of the information is stored in a database.
Preliminary results indicated that implantation was affected by the number and size of blastomeres on Day 3, and prediction of embryo implantation was superior to that of traditional manual scoring systems. Additional information about morphology and embryo development has also accumulated from the use of modern systems that allow time-lapse photography in combination with culture systems.
Aneuploidy Screening
Cytogenetic analysis of a biopsied polar body or blastomere has been used to screen embryos in order to detect those with an abnormal chromosome composition, a strategy known as aneuploidy screening or preimplantation genetic screening (PGS). The techniques employed for biopsy and diagnosis are described in subsequent blog posts, as well as the associated pros and cons. PGS continues to be a subject of considerable debate: time-lapse studies have demonstrated that removing a single blastomere has a negative effect on embryo development.
Follicular Indicators of Embryo Health
Each assisted conception cycle generates a number of waste products: luteinized granulosa cells, follicular fluids harvested from follicles at the time of oocyte retrieval and cumulus cells that can be removed and separated from the oocytes. These products have all been assayed to provide indexes of embryo developmental competence. Biomarkers quantified in serum and follicular fluid include cytokines, C-reactive protein and leptin, inhibin B and reactive oxygen species (ROS). Cumulus cell gene expression profiles have also been linked to the implantation potential of oocytes and embryos.
While many of these parameters are indicative of follicular differentiated status at the time of oocyte harvest, follicular fluid is a highly concentrated cellular exudate, which is accumulated over an extended period. Consequently, to date neither follicular fluid nor granulosa cell assays at the time of oocyte collection have provided a consistent measure for assessing the implantation potential of individual embryos. The degree of follicular vascularization and its relationship to mitochondrial segregation in embryonic blastomeres has also been promoted as a determinant of embryo developmental competence.
Secreted Factors
Assessment of products secreted by the embryo, the embryonic “secretome,” may be a better indicator of embryo development in vitro and in vivo than measurements of the follicular environment. For example, secretion of factors that regulate gamete transport and/or prepare the female tract for implantation has been used to predict embryo health. In this context, measurement of the amount of soluble human leukocyte antigen-G (HLA-G) into embryos culture media has been directly related to embryo quality and viability. mRNAs for HLA-G can be detected in human blastocysts, but the cellular origins and biology of soluble HLA-G are not clear.
The suitability of HLA-G as a predictor of embryo developmental potential has also been questioned, as the amount of HLA-G measured in embryo culture media appears to exceed the total protein content of the embryo itself.











Leave a Reply
Want to join the discussion?Feel free to contribute!